 
Acid chlorides are reacted with an alcohol to make an ester: Sterically. The name carboxyl comes from the fact that a carbonyl and a hydroxyl group are attached to the same carbon. Carboxylic acid derivatives are electrophilic at the carbonyl carbon. In these reactions the function of the acid is to produce a protonated alcohol. Primary alcohols and methanol react to form alkyl halides under acidic conditions by an S N 2 mechanism. 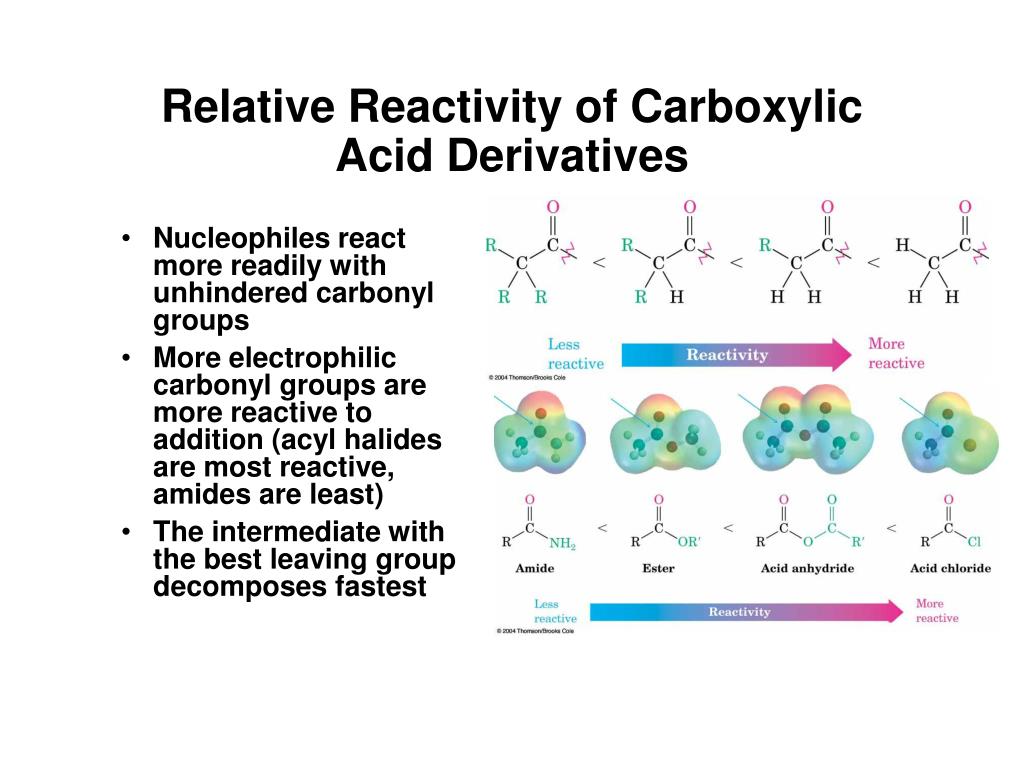
Not all acid-catalyzed conversions of alcohols to alkyl halides proceed through the formation of carbocations. Why were ortho-substituted compounds not included in the experiment?Ĭ. The importance of acetals as carbonyl derivatives lies chiefly in their stability and lack of reactivity in neutral to strongly basic environments. Carboxylic acids are organic compounds which incorporate a carboxyl functional group, CO 2 H. The overall result is an \(Sn1\) reaction. Why does one set of experiments give a positive $\rho$ value, whereas the other set of experiments gives a negative $\rho$ value?ī. The separation is simplified to an extent because the by-products are both gases. For example: CH3COOH+SOCl2 CH3COCl+SO2 +HCl C H 3 C O O H + S O C l 2 C H 3 C O C l + S O 2 + H C l. Summary This chapter contains sections titled: Reaction with Carboxylic Acids Reaction with Esters. (1) RCN + H 2: catalyst: RCHNH imine: H 2: RCH 2 NH 2 1º. Excess ammonia shifts the imine equilibrium to the left, as written below. 
(2nd equation), which would then add hydrogen to generate a 2º-amine. The $\rho$ value for the hydroxide ion-promoted hydrolysis of a series of meta-and para-substituted ethyl benzoates is +2.46 the $\rho$ value for amide formation for the reaction of a series of meta-and para-substituted anilines with benzoyl chloride is -2.78. It reacts with carboxylic acids to produce an acyl chloride, giving off sulfur dioxide and hydrogen chloride gases. An interesting correlation between the reactivity of carboxylic acid derivatives and their carbonyl stretching frequencies exists. The slope of a plot of the logarithm of the rate constant versus $\sigma$ is called the $\rho$ (ho) value. Electron-withdrawing substituents have positive $\sigma$ values the more strongly electron with drawing the substituent, the more positive its $\sigma$ value. The more electronegative leaving groups withdrawn electron density from the carbonyl, thereby, increasing its electrophilicity. $ Electron-donating substituents have negative $\sigma$ values: the more strongly electron donating the substituent, the more negative its $\sigma$ value. The relative reactivity of carboxylic acid derivatives toward nucleophile substitutions is related to the electronegative leaving group’s ability to activate the carbonyl. Information about the mechanism of the reaction undergone by a series of substituted benzenes can be obtained by plotting the logarithm of the observed rate constant determined at a particular pH against the Hammett substituent constant $(\sigma)$ for the particular substituent. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
